A Phase Change Materials Comparison: Vegetable-based vs. Paraffin-based PCMs
Source: Entropy Solutions
By Dr. William R. Sutterlin, chief science officer, Entropy Solutions
Passive processes for thermal energy storage have received a lot of attention in the past 25 years. These passive thermal energy storage materials can typically be divided into two parts, specific and latent. This paper will primarily focus on the concept of latent heat. However, a basic understanding of sensible heat is needed to fully appreciate latent heat. The concept of sensible heat is rather simple for most people to understand. In its most basic definition it is “thermal energy whose transfer to or from a substance results in a change in temperature.” 1
The most common example is the use of water to cool or heat another substance. If a relatively hot object is submerged into a relatively cool body of water, the hot object will cool by giving its thermal energy to the water and the water will warm. Water, the most commonly used sensible heat storage material, has a specific heat of 4.18 joules per gram per degree Celsius (J/(g·°C)). Therefore, if we want to heat 1 gram (approximately 1 cc or 1 ml) of water up 1 degree C, we have to put in 4.18 joules of energy into the water. Conversely, if we want to cool 1 gram of water down 1 degree C, we will take out 4.18 joules of energy.
However, if we want to freeze water by going from 1°C to 0°C, we will have to take out more than the normal 4.18 J/(g·°C) of specific heat. When water freezes it goes through a phase change, liquid to solid. The amount of energy that is removed in this change of phase is no longer 4.18 J/(g·°C) but 333 J/g. This relatively large amount of energy required to go through a change in phase between solids and liquids is referred to as the “latent heat of fusion.” Not only is this much energy needed to be removed to freeze water, but this much energy is also needed to be absorbed to melt 1 gram of ice. This is the reason why ice is such a good material to cool objects around it to 0°C.
It should also be noted that water goes through a phase change at 100°C at 1 atm. At these conditions water will boil, which is the process of going from a liquid to a gas. The amount of latent heat energy required to boil water is 2260 J/g. The energy required through this change in phase between liquid and gas is called the “latent heat of vaporization.” For this paper we will focus on the latent heat of fusion and those high latent heats associated with the phase change between solids to liquids and vice versa.
Phase change materials
Phase change materials
The term “phase change material” (PCM) is commonly used to describe materials that use phase changes (e.g., solidifying/liquefying) to absorb or release relatively large amounts of latent heat at relatively constant temperature. The most commonly used PCM is water/ice. Most people are familiar with using ice as a cooling agent at 0°C. Ice takes in a large quantity of heat when it melts (solid to liquid). Ice is very effective when used in a cooler to keep food or beverages at temperatures near water’s freezing point of 0ºC. However, the action of water solidifying (liquid to solid) and releasing heat at 0°C is also used. For example, farmers often spray their temperature-sensitive plants with water before a cold night when temperatures are expected to drop below 0°C. This is because the water will begin to freeze at 0°C and release 333 J/g and thereby assist in keeping the plant at 0°C and not dropping below 0°C where the plants would be damaged. Ice is an excellent PCM for maintaining temperatures at 0°C. However, ice is not an option if temperatures other than 0°C are desired.
1 Merriam-Webster Collegiate Dictionary. 11th Edition.
PCMs also find utility in pizza delivery. Materials undergoing a latent heat transformation at ~65ºC can be used to keep pizza warm. A wax-like PCM is melted at the pizza store before delivery. During delivery the wax-like PCM will transform from a liquid to a solid. This solidification is a phase change that releases heat. Therefore, the pizza will still be hot when it is delivered.
Other PCM applications include:
![]()
Figure 1. The thermal cycle of a PCM shows a temperature plateau effect at the PCM’s melting and freezing points. The effect of sensible heat in the solid and liquid can also be seen. Typically solids have lower specific heats than liquids.
Ideal characteristics of PCMs
Irreversible polymorphism and supercooling: Irreversible heat transfer generally adversely affects performance. A large temperature difference between the melting and freezing process will typically translate to an overall decrease in efficiency for the entire process. Little or no supercooling is desired. The melt temperature and the freeze temperature should be in close proximity to each other. The presence of polymorphism can result in two or more endothermic or exothermic peaks, which is undesired in most applications. However, this could be advantageous in certain thermo-engineering applications.
Ice and gel packs have become extremely popular for keeping materials cold around 0°C. These low-cost devices have several advantages. They perform well and are non-toxic, non-flammable, environmentally friendly and easy to use. The sole disadvantage: They are useful only in applications requiring a temperature of 0°C. If one wishes to obtain a water-based PCM lower than 0°C, a salt can be added to the water to lower the freezing point. However, this significantly decreases the latent heat and broadens the melt/freeze temperature. The technique is familiar to anyone who has added salt to the ice used in making homemade ice cream or seen salt applied to icy roads.
Similarly gel packs are types of salts. The predominant compound in many gel packs is sodium polyacrylate, which is also commonly used in disposable diapers. The addition of sodium polyacrylate, however, changes the crystal structure of ice and diminishes its effectiveness as a PCM.
Salt hydrates
Salt hydrate PCMs have several problems related to practical applications. Foremost is the limited temperature ranges available to salts in meeting specific temperature needs. Few salts melt between 1 and 150°C. Eutectic mixtures and salt hydrates are pursued for these missing temperature ranges.
Other PCM applications include:
- Anti-icing systems for bridges
- Socks that keep feet warm in winter
- Vests that keep outdoor workers cool in summer
- Containers that keep foods, beverages and medicines at the right temperature for longer periods during shipping
- Building materials that keep living spaces comfortable
- Packaging that protects temperature-sensitive medical devices and other electronics
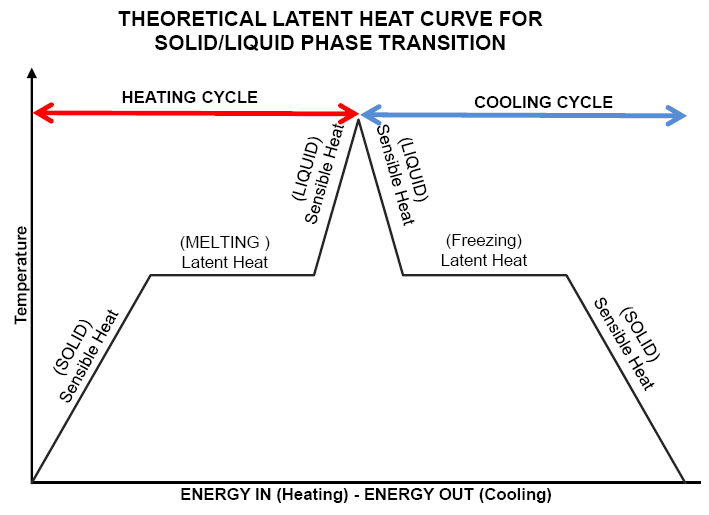
Figure 1. The thermal cycle of a PCM shows a temperature plateau effect at the PCM’s melting and freezing points. The effect of sensible heat in the solid and liquid can also be seen. Typically solids have lower specific heats than liquids.
Ideal characteristics of PCMs
The value of a PCM chemical is measured by the following performance measures:
Latent heat: Preferred latent heats are greater than 160 J/g and more preferably >180 J/g. A high latent heat of fusion, measured in joules per gram (J/g), means that a lesser amount of material is needed to store a given amount of energy.
Sharpness of latent heat release and absorption: To a first approximation, the release and absorption of latent heat over a narrow temperature range is an important performance criteria. However, for some applications, broad heat release rates of up to 10 or even 30°C will not adversely affect performance in a properly engineered consumer device.
Irreversible polymorphism and supercooling: Irreversible heat transfer generally adversely affects performance. A large temperature difference between the melting and freezing process will typically translate to an overall decrease in efficiency for the entire process. Little or no supercooling is desired. The melt temperature and the freeze temperature should be in close proximity to each other. The presence of polymorphism can result in two or more endothermic or exothermic peaks, which is undesired in most applications. However, this could be advantageous in certain thermo-engineering applications.
Stability to thermal cycling and aging: Over the course of hundreds or thousands of freezes and melts the phase change transition temperature and latent heat energies should remain consistent. Changes in the hydration sphere of some PCM molecules over time can affect the melt and freeze points and their respective latent heats. This is the primary reason that one of the most-used stability tests for PCMs is the thermal cycle test that involves multiple melt and freeze cycles.
Final encapsulation or containment: Plastic tends to be the least expensive container for PCM chemicals. However, plastics suffer from low thermal conductivity when packaged in containers with thick plastic walls. Various metals can be used for PCM encapsulation to increase thermal conductivity. However, metals tend to be more costly and will corrode with various PCMs. Careful consideration must be given to the compatibility of the individual PCM with plastic or metal chosen for the application. The final packaging and containment of a PCM is a factor that should be given considerable thought and planning and not considered as an afterthought.
Safe to use: The PCM should be environmentally friendly and non-poisonous, non-flammable and non-explosive.
Cost effectiveness: The useful heat in joules per gram of phase change material is a primary performance criteria for PCM chemicals. On a cost basis, a minimum in dollars per joule of useful heat is desired.
Temperature: Applications are typically temperature-specific. The temperature desired is often within 1 or 2 degrees. Therefore, 200 different PCMs would be needed to target each degree between -50°C and 150°C.
Types of PCMs
Water and gel packs
Types of PCMs
Water and gel packs
Ice and gel packs have become extremely popular for keeping materials cold around 0°C. These low-cost devices have several advantages. They perform well and are non-toxic, non-flammable, environmentally friendly and easy to use. The sole disadvantage: They are useful only in applications requiring a temperature of 0°C. If one wishes to obtain a water-based PCM lower than 0°C, a salt can be added to the water to lower the freezing point. However, this significantly decreases the latent heat and broadens the melt/freeze temperature. The technique is familiar to anyone who has added salt to the ice used in making homemade ice cream or seen salt applied to icy roads.
Similarly gel packs are types of salts. The predominant compound in many gel packs is sodium polyacrylate, which is also commonly used in disposable diapers. The addition of sodium polyacrylate, however, changes the crystal structure of ice and diminishes its effectiveness as a PCM.
Salt hydrates
Salt hydrates as PCMs have been among the most-researched latent heat storage materials. Salt hydrates are often the lowest-cost PCM behind water and gel packs. Numerous trials and subscale tests have been carried out on these compounds. The material comprises M·nH2O where M is an inorganic compound.
Salt hydrate PCMs have several problems related to practical applications. Foremost is the limited temperature ranges available to salts in meeting specific temperature needs. Few salts melt between 1 and 150°C. Eutectic mixtures and salt hydrates are pursued for these missing temperature ranges.
Another significant problem, A. Abhat noted in 1983, “is that most of them melt incongruently, i.e. they melt to a saturated aqueous phase and a solid phase which is generally a lower hydrate of the same salt. Due to density differences, the salt phase settles out and collects at the bottom of the container, a phenomenon called decomposition. Unless specific measures are taken, this phenomenon is irreversible, i.e. during freezing, the solid phase does not combine with the saturated solution to form the original salt hydrate.” 2 Abhat’s observation was repeated by Carl Verner (“Phase Change Thermal Energy Storage,” 1997) 3 and G.A. Lane ("Phase Change Material for Energy Storage Nucleation to Prevent Supercooling," 1991) 4.
Salt hydrates also have poor nucleating properties that result in what is termed supercooling of the liquid salt hydrate prior to freezing. This must be overcome by the addition of a suitable nucleating agent that has a crystal structure similar to that of the parent substance.
Other issues of using salt hydrates involve the volume change, the corrosive nature of the salt hydrate and the toxicity of some of these materials. Often the volume change in the solid/liquid phase change of a salt hydrate is up to 10 percent. While this is significant, it can be accommodated in special packaging. The packaging needs to be specific to the type of salt hydrate used. Many salt hydrates are corrosive to metals. The toxicity of salt hydrates also varies widely. Therefore, the safety data sheets should be carefully checked for human toxicity and environmental damage before disposal.
Paraffins
Paraffins
Paraffins are high molecular mass hydrocarbons with a waxy consistency at room temperature. Paraffins are made up of straight chain hydrocarbons. The paraffins are separated into two main subgroups, straight chained (n-Paraffin) and branched chained (iso-Paraffin). Typically iso-paraffins do not make good PCMs because of steric hindrances in their molecular packing. The presence of any iso-paraffins in a PCM can severely degrade the thermal performance of the PCM. Therefore we will only focus on the n-paraffins.
The melting point of paraffins is directly related to the number of carbon atoms within the material structure, with alkanes containing 12-40 C-atoms possessing melting points between 6 and 80 degrees Celsius. These are termed pure paraffins and should not be confused with paraffin waxes. Paraffin waxes contain a mixture of hydrocarbon molecules with various carbon numbers with lower melting points and poorer latent heats than pure paraffins. Paraffin waxes are often considered a low-grade PCM.
Paraffins form a good PCM candidate for certain applications and temperature ranges. Paraffins have good thermal storage capacity and are proven to freeze without supercooling. Paraffins also have the advantages of chemical stability over many heating and freezing cycles 5 and high heat of fusion. They are non-corrosive, compatible with most materials and non-reactive to most materials of encapsulation.
Some disadvantages are associated with paraffins. Paraffin products are often reported to have very high latent heats. However, the details are most important in this assessment. Commercially cost-effective paraffins are mixtures of alkanes and therefore do not have well-defined melting points. For example, 98% hexadecane has a latent heat of about 230 J/g at a melting point of 18°C, but due to the very small amount of hexadecane in petroleum crude oil (a small fraction of 1%) and the enormous separation costs (repeated high-vacuum multi-stage distillation) to achieve a 98% pure product, hexadecane is not a viable commercial product as exemplified by the price of over $10 per pound. Wax products are commercially viable for a variety of applications including uses in canning and candle making. A C20-C24 canning wax has a latent heat of 150 J/g and melts over a 7°C temperature range— considerably poorer performance than +98% n-paraffin products found in most literature. Pure paraffins are also limited in the range of melting points they can target. Many researchers try to expand this temperature range by creating mixtures of two or more paraffins. This, however, has the detrimental effect of broadening the melt/freeze temperature and lowering the latent heats even more.
Other concerns with paraffins used as PCMs are social dynamics. Paraffins are made from petroleum products, which increases reliance on crude oil. Paraffin prices have followed the unstable price of petroleum. Furthermore, petroleum-derived paraffins have geopolitical consequences and contribute to the increase in carbon emissions blamed for global warming.
Vegetable-based PCMs
Vegetable-based PCMs
In the past, the phase change material market has been dominated by paraffin products and salt hydrates. Paraffins have become more popular than the salt hydrates in the PCM market. However, vegetable compounds are making strong inroads. The ability of vegetable compounds to compete in this market depends on their price/performance characteristics relative to paraffins.
The Department of Agriculture and the National Science Foundation have sponsored research to investigate the potential for vegetable-derived compounds to become a significant factor in the PCM market.6, 7, 8 During these investigations, researchers were able to produce around 300 different fat- and vegetable-oil-based PCMs ranging from minus -90°C to 150°C with latent heats between 150 and 220 J/g.
The safety, environmental and social benefits of using vegetable-based PCMs are significantly greater than those of paraffins. Paraffins can be toxic and have laxative effects when ingested. Many vegetable-based PCMs can be considered “food grade” or have no effect when ingested. Researchers under the Department of Agriculture research program also discovered that many vegetable-based PCMs had higher flash points and 10-20 percent longer horizontal flame propagation rates than did their temperature-comparable paraffin-based counterparts.
Paraffins are long chain alkanes. This means that there is no “active” site on the paraffin molecule for microbes and bacteria to begin their breakdown. While this is attractive for the life of the PCM, it presents environment problems when the paraffin is discarded in a landfill. The paraffin would stay undegraded for decades. Many vegetable-based PCMs have shown stability after 30 years of accelerated aging tests. 9 However, these same vegetable-based PCMs will degrade in six months or less when discarded in a landfill. Many vegetable-based PCMs can be derived locally using common agricultural crops. This leads to a more stable price and regionally based feedstocks. Vegetable-based PCMs are considered to be nearly carbon neutral as recently ranked by the USDA BioPreferred program. 10
Vegetable-based PCMs are able to provide price/performance characteristics superior to paraffin PCM chemicals because 1) the natural vegetable-based feedstocks are available at relatively low prices and 2) multiple reaction-modification schemes provide many degrees of freedom to produce a variety of premium products. These vegetable-derived chemical building blocks are available at concentrations considerably greater than any particular n-paraffin is available in crude oil. Because of this, vegetable-based PCMs have a distinct competitive advantage over paraffins and could corner the PCM market.
Vegetable-based PCMs are able to provide a price/performance characteristic superior to salt hydrate PCM chemicals over most of the temperatures between -90°C and 150°C because viable salts or salt hydrates simply are not available over much of that temperature range. For the limited temperatures where viable salt PCM chemicals are available, vegetable-based PCMs have advantages of 1) being renewable and more environmentally friendly and 2) being compatible with wall boards or similar materials that would absorb water from hydrates and render the salt hydrates ineffective as PCM chemicals. 3) Vegetable-based PCMs are capable of being microencapsulated; salt hydrates are not.
Summary
Summary
In a head-to-head comparison, phase change materials made from vegetable sources have the edge on paraffin-based PCMs. 1) Vegetable-based PCMs are made from renewable “farmer-made” products. Paraffins are made from petroleum products. 2) Many vegetable-based PCMs have equal or higher latent heats associated with them, typically around 200 J/g. 3) Vegetable-based PCMs are able to target more temperature ranges with more product varieties (around 300) than paraffins (around eight). 4) Many vegetable-based PCMs have higher flash points, longer ignition rates and longer horizontal flame propagation rates than paraffins. 5) Vegetable-based PCMs are biodegradable. Paraffins are not. 6) Most importantly, many fat- and vegetable-based PCMs sell at significantly
lower prices than paraffin-based PCMs.
References
1 Merriam-Webster Collegiate Dictionary. 11th Edition.
2 Abhat, A. “Low Temperature Latent Heat Thermal Energy Storage: Heat Storage Materials,” Solar Energy, Vol. 30, No. 4, 313-332, 1983.
3 Verner, C. Phase Change Thermal Energy Storage. Thesis, Brighton University (See http://freespace.virgin.net/m.eckert/carl_vener's_dissertation.htm), May 1997.
4 Lane, G.A. “Phase Change Material for Energy Storage Nucleation to Prevent Supercooling,” Solar Energy Materials and Solar Cells, 27 135-160, p4, 1991.
5 S.D. Sharma, D. Buddhi, R. L. Sawhney, "Accelerated Thermal Cycle Test of Latent Heat Thermal Storage Material," Solar Energy, 66, (1999), 483.
6 National Science Foundation, SBIR, Award Number - 0750470
7 U.S. Department of Agriculture, SBIR, Project Number MOK-2003-05519
8 U.S. Department of Agriculture, NRI, Project Number MOR-2005-02692
9 Internal Studies Conducted by Phase Change Energy Solutions, 2009; Entropy Solutions Inc., 2009; University of Missouri-Columbia, 2007.
10 www.biopreferred.gov
This website uses cookies to ensure you get the best experience on our website. Learn more
